San Francisco and the broader Bay Area have long been the epicenter of life sciences and medical technology innovation. As a premier Ligating Clips and Appliers Manufacturer and Exporter in San Francisco, we leverage the unique ecosystem of the "Biotech Bay." This region is home to world-class research institutions like UCSF and Stanford, which drive the demand for high-precision minimally invasive surgical (MIS) instruments.
Our manufacturing processes utilize AI-driven quality control, ensuring each ligating clip meets the rigorous standards required by California's top trauma centers.
Located near the Port of San Francisco and SFO International Airport, we offer streamlined export services to Asia, Europe, and South America.
Adhering to FDA and CE standards, our products are designed for the high-compliance environment of the US West Coast healthcare market.
The industrial landscape in San Francisco is shifting toward "Smart Manufacturing." By integrating AI and advanced robotics into the production of polymer and titanium ligating clips, we provide surgeons with tactile feedback and secure vessel occlusion that was previously unavailable. This localization of high-tech production ensures that San Francisco remains a leader in the global medical device export market.
Sqm Production Base
Core Patents
Years R&D Exp
Quality Inspected
A High-Tech Enterprise Integrating R&D, Manufacturing, Sales, and Services
Zhejiang Centuri Medical Co., Ltd., is a high-tech enterprise that integrates research and development, manufacturing, sales, and after-sales services. The company is committed to innovation and production in the field of minimally invasive surgical instruments and medical products, providing advanced medical solutions for the global market.

Zhejiang Centuri Medical Co., Ltd. has established a research and production base of 20,000 square meters, which includes a 5,000-square-meter cleanroom and a 500-square-meter laboratory. The company is equipped with a comprehensive quality management system, advanced manufacturing equipment, and highly automated production and testing processes, ensuring that every product meets international standards.
The company has successfully passed the "Medical Device Production Quality Control" and "Management Standard" system evaluations and has obtained ISO 13485 certification for the medical device quality management system. Our product portfolio includes disposable puncture devices, digital endoscopes, electrocautery surgical instruments, surgical staplers, and endoscopic imaging systems, all of which have obtained domestic registration certificates and EU CE certification.
In the field of minimally invasive surgical instruments, we boast a highly experienced R&D team and maintain long-term collaborations with renowned hospitals and clinical experts. To date, the company has obtained more than 50 core product patents, including both invention and utility model patents.
Modern surgery in San Francisco clinics is moving away from metal towards Non-Absorbable Polymer Clips, which offer MRI compatibility and zero interference with CT scans.
Appliers now feature 20-degree and 45-degree angled tips to facilitate access in complex robotic-assisted surgeries (RAS), a growing trend in California hospitals.
Medical exporters are increasingly focusing on biodegradable packaging and recyclable instrument components to align with San Francisco's green initiatives.
The integration of ligation technology with digital imaging systems allows for real-time verification of clip placement. As a leading manufacturer, we are developing "Smart Appliers" that signal to the surgeon when the optimal closing pressure has been reached, minimizing the risk of vessel damage during delicate procedures.
Our ligating clips and appliers are specifically optimized for the following clinical scenarios prevalent in the Northern California medical network:
Over 10 years of experience in surgical instruments R&D, providing comprehensive market analysis and customization.
Strong regulatory affairs team assisting in local registration to ensure products reach the market quickly.
Continuous technical support, product training, and global warranty services for seamless usage.
Flexible production arrangements and priority delivery to meet urgent customer demands.
Carefully selected shipping channels to ensure fast and reliable delivery to your doorstep.
Best prices by eliminating middleman markups, ensuring clients enjoy the best value for money.
We offer OEM, ODM, custom brand design, and marketing planning services to help clients build their brands and expand their market presence. Our development strategy includes comprehensive distribution plans and marketing support for our global partners.
 Disposable Ligating Clips for Endoscopic Use - Tiga-Lok Appliers Set
Disposable Ligating Clips for Endoscopic Use - Tiga-Lok Appliers Set
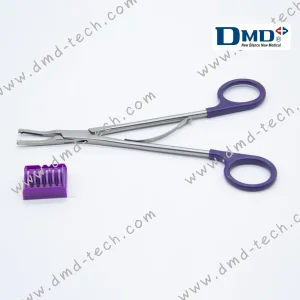 Reusable Large Polymer Hemolock Ligating Clips Open Appliers, 20cm
Reusable Large Polymer Hemolock Ligating Clips Open Appliers, 20cm
 Premium Polymer Ligating Clips for Standard Clip Appliers
Premium Polymer Ligating Clips for Standard Clip Appliers
 Tiga-Lok Disposable Ligating Clips - Complete Set Size Large Yellow
Tiga-Lok Disposable Ligating Clips - Complete Set Size Large Yellow
 Size Small Endoscopic Ligating Clips Set with Tiga-Lok Appliers - 6PCS
Size Small Endoscopic Ligating Clips Set with Tiga-Lok Appliers - 6PCS
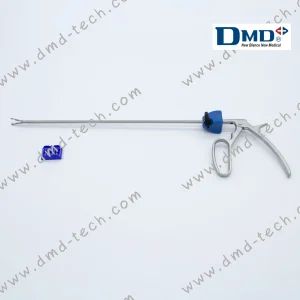 Poly-Lok Endo Appliers Customized Angle for Polymer Ligating Clips
Poly-Lok Endo Appliers Customized Angle for Polymer Ligating Clips
 Endoscopic Ligating Clips Set with Disposable Tiga-Lok Appliers - Variety
Endoscopic Ligating Clips Set with Disposable Tiga-Lok Appliers - Variety
 Disposable Tiga-Lok Ligating Clips for Endoscopic Appliers - Colorful Set
Disposable Tiga-Lok Ligating Clips for Endoscopic Appliers - Colorful Set
Zhejiang Centuri Medical Co., Ltd. is always customer-oriented, focusing on technological innovation, high-quality products, and attentive after-sales service. With a strong foundation in the domestic market and a strategic export presence in San Francisco, we are actively expanding into international markets and contributing to global healthcare. We look forward to collaborating with you and advancing medical technology together for the betterment of human health.
Website Development and Product Support: We offer high-quality product images and videos to help our customers showcase their products on their websites, enhancing brand visibility and market competitiveness. If you have any website development or marketing needs, Centuri Medical is your trusted partner.