














| Customization: | Available |
|---|---|
| Type: | Basic Surgical Instruments Sets |
| Material: | TPU |
| Model NO. | WPQA250/260-250/250 | Sterilization | Ethylene Oxide (EOS) |
| Quality Guarantee | Two Years | Group | Adult |
| Certification | CE / ISO 13485 | Safety Standard | 93/42/EEC |
| Feature | Eco-Friendly | Classification | Class II |
| Packing | Blister (1 PCS/Box) | Shelf Life | 3 Years |
Prepare the surgical site according to standard procedure, making sure the skin is clean and dry.

Make an incision along the marked incision line
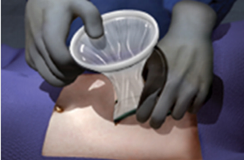
Insert the wound protector-retractor's green ring into the operative site

Carefully check to ensure that no bowel or tissue entrapment has occurred

Pull up the white retraction ring and flip the outer ring until desired retraction is achieved
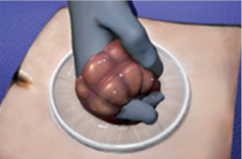
Adjust the insert ring within the operative site for optimal placement
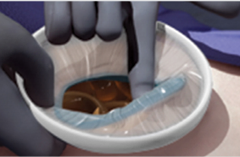
Retrieve by simply removing the green inner ring from the peritoneal cavity


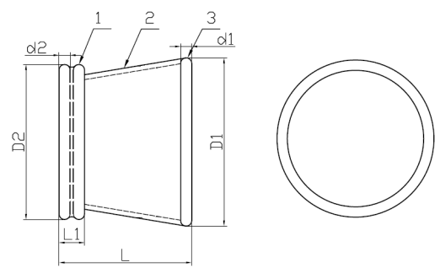
Model: WPQA (Material: TPU)
1. Outer Ring 2. Channel 3. Insert Ring
Model: WPQB (Material: Silicone)
1. Insert Ring 2. Channel 3. Outer Ring
| Property | Specifications / Performance |
|---|---|
| Product Name | Disposable Wounder protector/retractor |
| Appearance | Smooth, soft, clear outline, no burrs or scratches, free from damage or contamination. |
| Strength | Model WPQA channel heat sealing strength ≥ 0.140 N/mm. |
| Elasticity | Rings produce elastic deformation under 20 N radial force and recover after release. |
| Adjustability | Channel length adjustable; outer ring can be rolled down or extended manually. |
| Sterility | Ethylene oxide sterilized; residual EO ≤ 10μg/g. |
| Model | Specification | Outer D(mm) | Insert D(mm) | Channel L(mm) |
|---|---|---|---|---|
| WPQA | WPQA40/40-40/100 | 40 | 40 | 100 |
| WPQA | WPQA60/70-60/150 | 60 | 70 | 150 |
| WPQA | WPQA100/100-100/150 | 100 | 100 | 150 |
| WPQA | WPQA150/160-150/250 | 150 | 160 | 250 |
| WPQA | WPQA250/260-250/250 | 250 | 260 | 250 |
| WPQB | WPQB40/30-15/25 | 40 | 30 | 25 |
| WPQB | WPQB70/70-35/35 | 70 | 70 | 35 |

Leadership with over 10 years of experience in global medical device quality management and product registration (CE, ISO, FDA 510K).
Direct cooperation with factories for wholesale pricing. We support manufacturers in maintaining CE/ISO certifications.
Professional team in contact with global medical experts, keeping up with the latest industry technologies and knowledge.


 Centuri Medical
Centuri Medical