

1 / 5










| Still deciding? Get samples of $ ! US$ 10/Piece Request Sample |
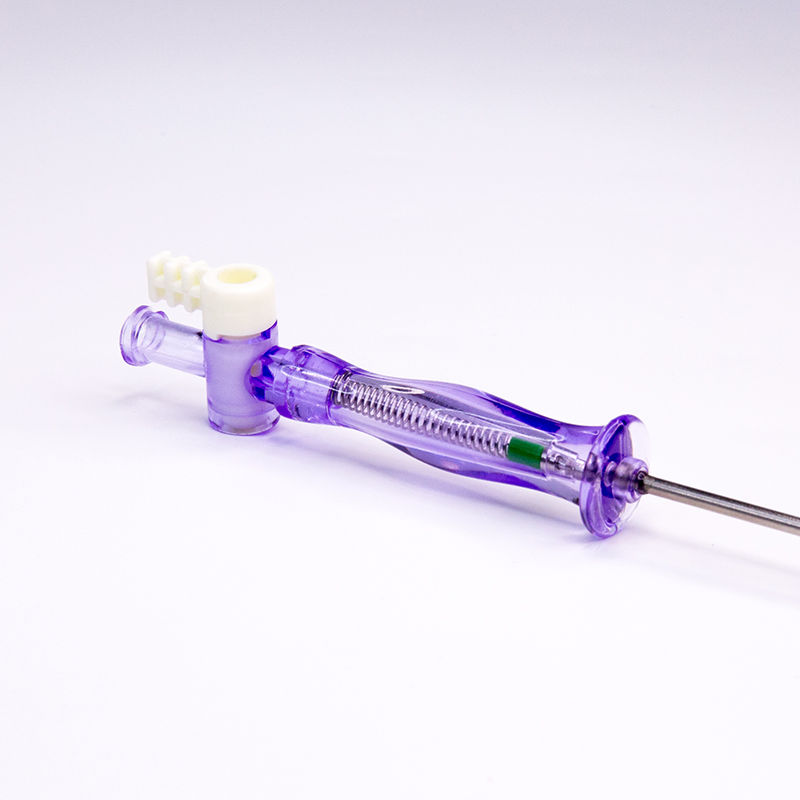
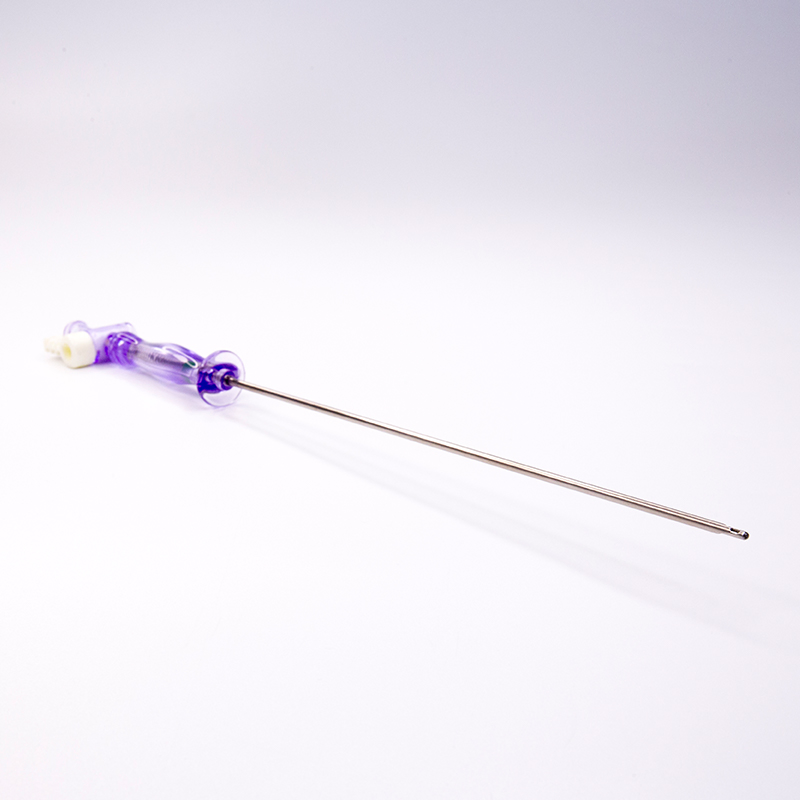
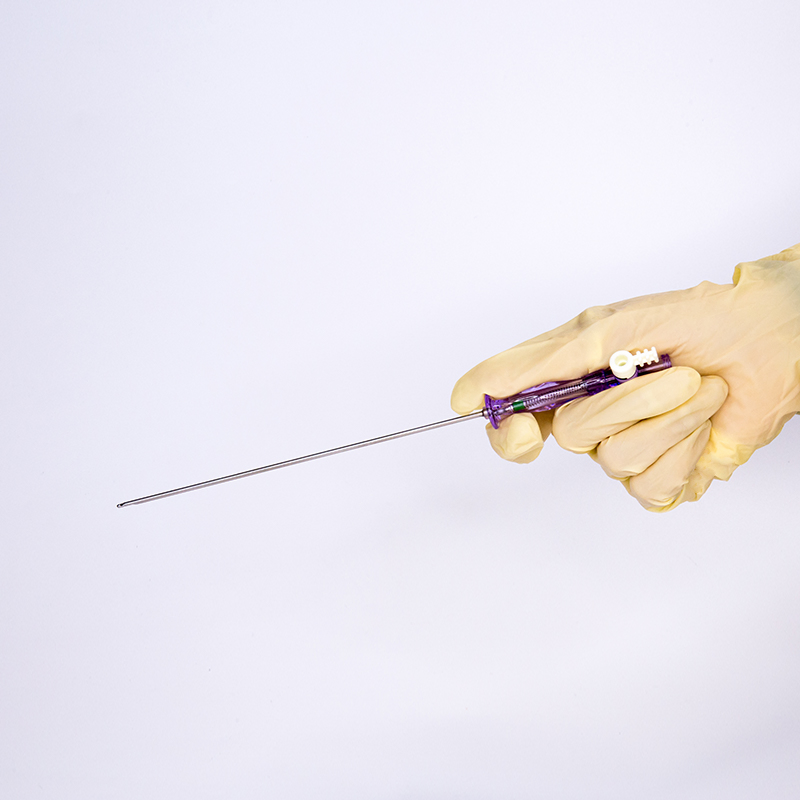
FDA 510K cleared laparoscopic Pneumoneedles Veress Needles surgical Instruments. High-quality medical devices manufactured with more than 17 years of R&D and manufacturing expertise. These needles are exported to more than 40 countries and utilized in over 1000 hospitals worldwide.
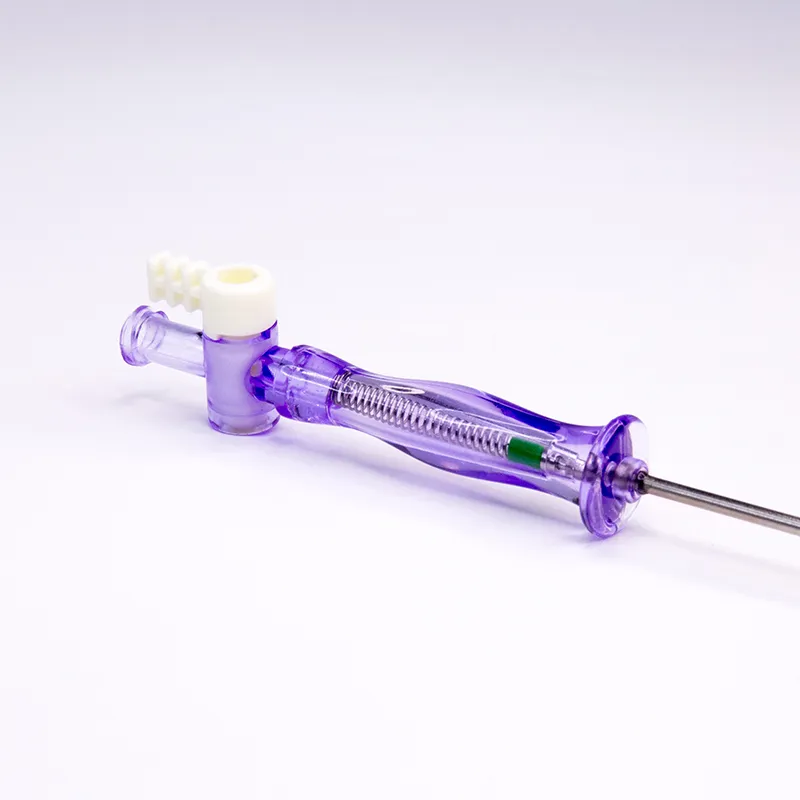



The veress needles have been widely adopted in European and American markets, including the USA, UK, France, Germany, Italy, Belgium, Spain, Japan, and South Korea.
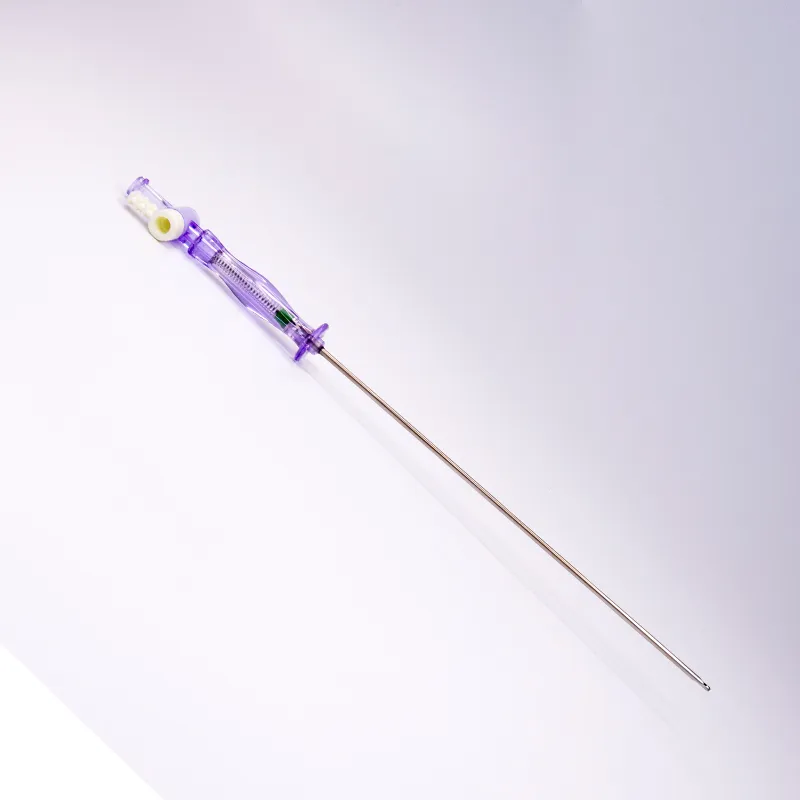

| Product Code | Working Length (mm) |
|---|---|
| VN-120 | 120 |
| VN-150 | 150 |
| VN-120L | 120 |
| VN-150L | 150 |


 Centuri Medical
Centuri Medical