




1 / 5










| Still deciding? Get samples of $ ! US$ 20/Piece Request Sample |
Endoscopic instruments Retrieval bags for laparoscopic surgical incision for Cholecystectomy
A hi-tech medical devices manufacturer of more than 19 years R&D and manufacturing expertise. These veress needles are exported to more than 40 countries and used in more than 1000 hospitals worldwide.



Passed GMP of NMPA (CHINA FDA) and US FDA factory registration and ISO13485:2016 medical devices quality management system.


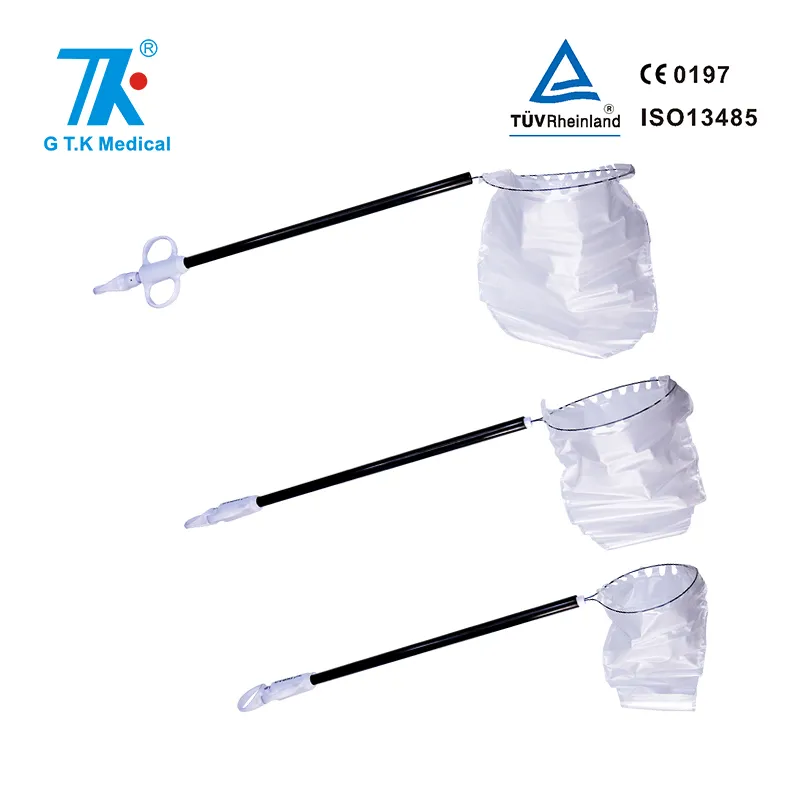

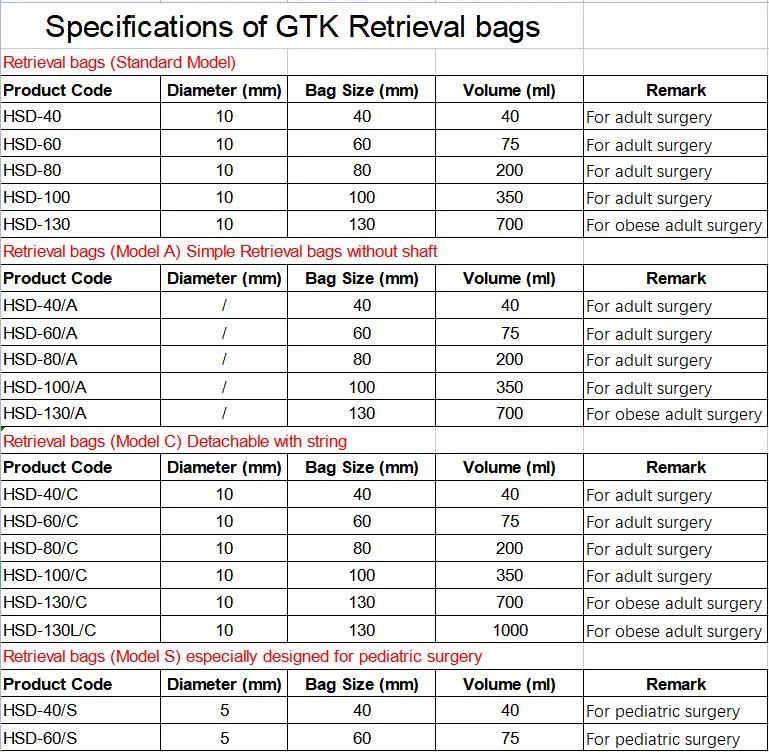
Applied more than 700 Patents at home and abroad, including more than 30 PCT Patents.

 Centuri Medical
Centuri Medical