














| Customization: | Available |
|---|---|
| Material: | Silicone |
| Ethylene Oxide Sterilization: | Ethylene Oxide Sterilization |
Prepare the surgical site according to standard procedure, making sure the skin is clean and dry.

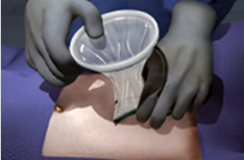


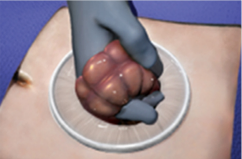
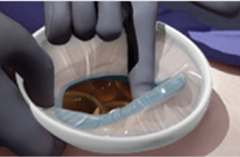
Model: WPQA (Material: TPU)
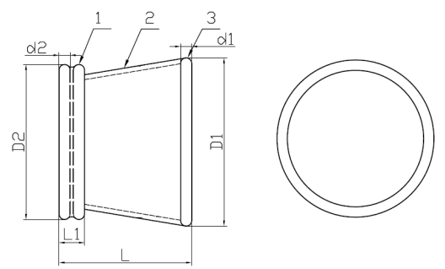
1. Outer Ring 2. Channel 3. Insert Ring
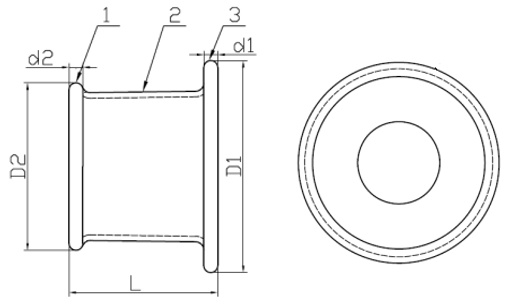
Model: WPQB (Material: Silicone)
1. Insert Ring 2. Channel 3. Outer Ring
| Item | Standard Performance |
|---|---|
| Product Name | Disposable Wounder protector/retractor |
| Appearance | Smooth, soft, no burrs, scratches or defects; clear outline. |
| Seal Strength | WPQA channel heat sealing strength ≥ 0.140 N/mm. |
| Elasticity | Rings recover elastically under 20 N radial force. |
| Adjustability | Channel length is adjustable; outer ring rolls down via manual rotation. |
| Sterility | Sterilized with Ethylene Oxide (EO); Residual EO ≤ 10μg/g. |
1. Regulatory Expertise: Led by experts with over 10 years of experience in medical device quality management and global product registration (FDA 510K, CE, ANVISA).
2. Direct Manufacturing Links: Strong partnerships with certified Chinese factories (CE/ISO/FDA). Direct factory wholesale pricing through strategic investments.
3. R&D and Quality Control: Constant collaboration with global medical experts to incorporate the latest industry technologies and maintain high quality standards.


Standard Aseptic Blister Packaging
 Centuri Medical
Centuri Medical