




1 / 5










| Customization: | Available |
|---|---|
| Material: | Silicone |
| Ethylene Oxide Sterilization: | Ethylene Oxide Sterilization |
Prepare the surgical site according to standard procedure, making sure the skin is clean and dry.

Make an incision along the marked incision line
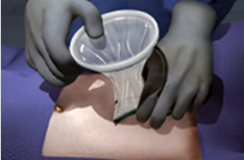
Insert the wound protector-retractor's green ring into the operative site

Ensure no bowel or tissue entrapment has occurred before retraction

Pull retraction ring until the green ring sits tightly; flip outer ring for desired retraction
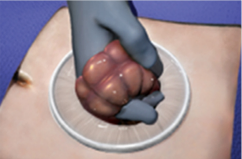
Ensure stable placement within the operative site
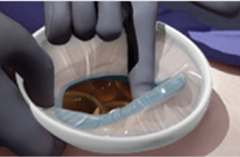
Retrieve by removing the green inner ring from the peritoneal cavity

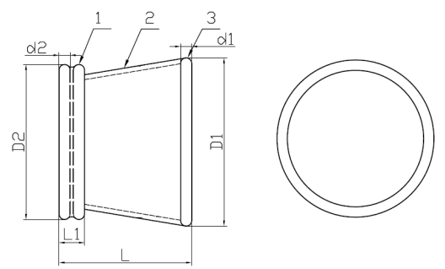
Model: WPQA (Material: TPU)
1. Outer Ring | 2. Channel | 3. Insert Ring
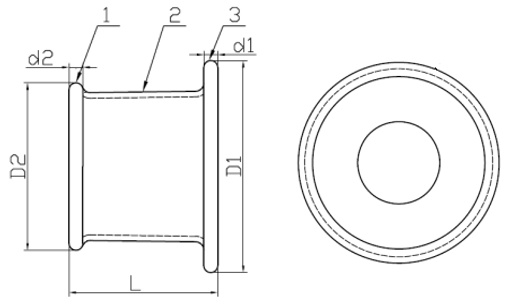
Model: WPQB (Material: Silicone)
1. Insert Ring | 2. Channel | 3. Outer Ring
| Product Name | Disposable Wound protector/retractor |
| Appearance | Smooth, soft, clear outline, no burr, no scratches or shrinkage defects. |
| Performance | WPQA channel heat sealing strength ≥ 0.140 N/mm. Elastic deformation allows recovery under 20 N radial force. |
| Package | Aseptic packaging, peel strength ≥ 0.1 N/mm, uniform sealing without tearing. |
| Sterility | Ethylene oxide (EO) sterilized; residual EO ≤ 10μg/g. |


 Centuri Medical
Centuri Medical